Genomics
Institute
What is CRISPR?
A brief introduction to CRISPR genome editing
Jennifer Doudna Introduces CRISPR
In her first TED Talk back in 2015, IGI Founder Jennifer Doudna gives a great introduction to how the CRISPR-Cas9 system lets scientists edit DNA in almost any kind of cell or organism. This revolutionary genetic engineering tool has created monumental opportunities to cure genetic disease, improve agriculture, and foster a healthy environment — and simultaneously forces us to grapple with difficult ethical dilemmas.
How CRISPR Works
DNA is the instruction manual for life on Earth. It encodes the fundamental properties of an organism — how it lives, grows, and reproduces. Changing a DNA sequence in a living cell is known as genome editing or gene editing. For a long time, this was either impossible or extremely challenging.
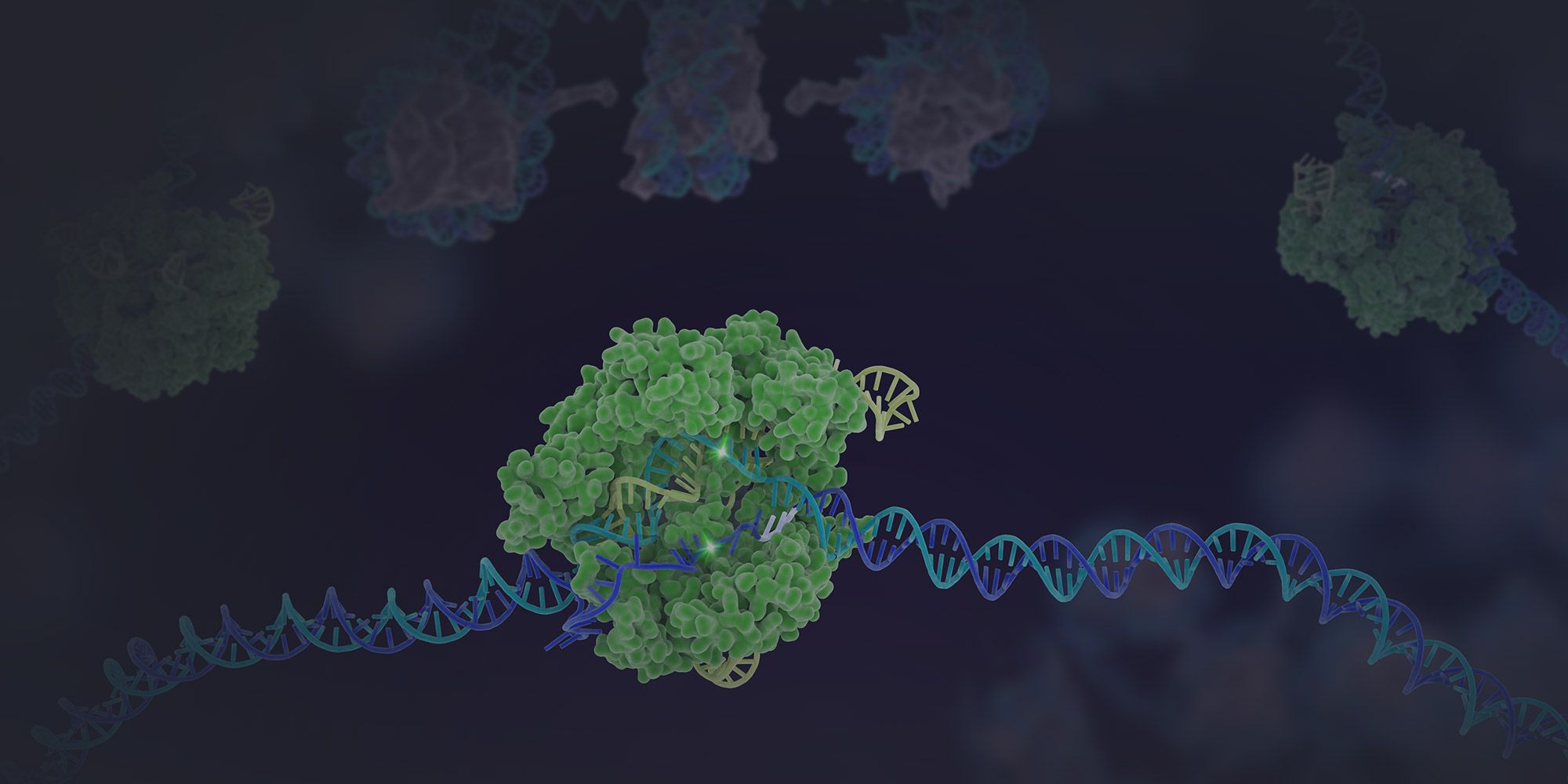
The development of CRISPR genome editing has revolutionized scientists' ability to edit the genome. In 2012, research by IGI founder Jennifer Doudna, Emmanuelle Charpentier, and their teams developed a method of repurposing a bacterial immune system called CRISPR — an acronym that stands for Clustered Regularly Interspaced Short Palindromic Repeats — to make breaks in DNA at precise locations, using a CRISPR-associated protein (Cas9) like molecular scissors to cut DNA. When DNA is cut, this creates an opportunity to change the DNA sequence at and around the cut site by harnessing the cell's natural repair pathways.
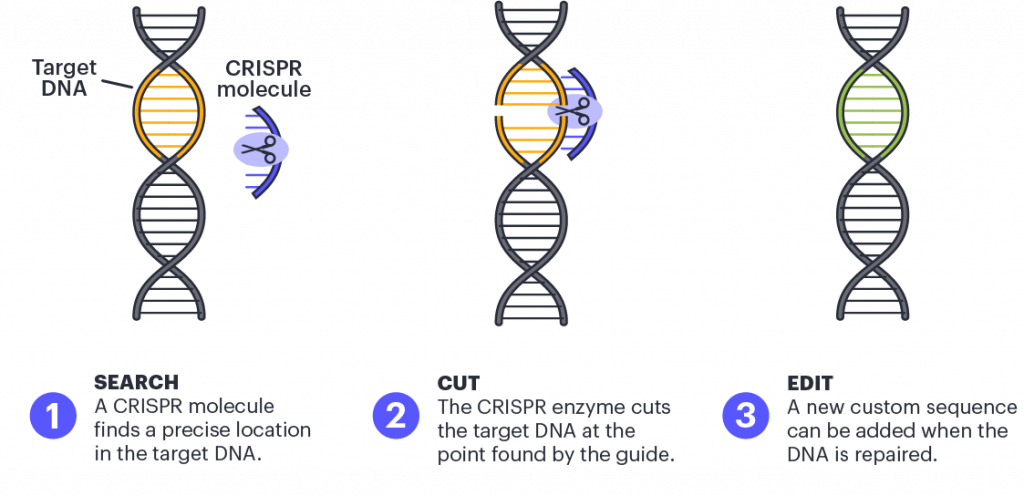
Scientists can edit the genome of living organisms by adding new fragments of DNA for the cell to use as a template when it repairs the break in the DNA. In this way, scientists can replace a disease-causing mutation with a healthy sequence or make other modifications to the genome.
Scientists can also use CRISPR to “knock out” a gene entirely, disrupting its function or causing a cell to stop making the protein. This is the technique that is used in Casgevy, the first approved CRISPR therapy for sickle cell disease. It is also possible modify how genes are expressed with "epigenetic editing," which can dial up or down the effect of a gene, often in a temporary way. The CRISPR toolbox is continually growing with newer approaches like base editing and prime editing that allow scientists to change single letters in the genetic code.
Together, these methods of genome editing give scientists powerful new tools to treat disease, improve agriculture, develop new approaches to stopping and adapting to climate change, and study fundamental questions of biology by having the ability to edit the DNA of almost any organism.
Learn More About CRISPR
Dive deeper with our learning resources for everyone from beginners to advanced learners.
CRISPR History

In this WNYC Radiolab episode, Jad Abumrad and Robert Krulwich explore the development of CRISPR with Jennifer Doudna, Eugene V. Koonin, Beth Shapiro, and Carl Zimmer.
"Hidden inside some of the world’s smallest organisms is one of the most powerful tools scientists have ever stumbled across. It's a defense system that has existed in bacteria for millions of years and it may some day let us change the course of human evolution."
How CRISPR Is Changing Medicine & Agriculture
CRISPR genome editing has quickly been adopted in scientific research labs, medicine, and agriculture. Learn more in our reader-friendly deep dives into how genome editing is affecting the world we live in today.
CRISPR Ethics & Access

Powerful technologies like CRISPR raise many ethical questions. These range from how the technology should or should not be used to who has access to the technology and the benefits it may bring. At IGI, we are committed to guiding responsible use of CRISPR technology, working with stakeholders and policymakers around the world, and creating new models for access and affordability for advances in medicine and agriculture. Visit our Public Impact program page to learn more about IGI's work in CRISPR regulation, access, and affordability.
"All of us have a huge responsibility, to consider carefully both the unintended consequences as well as the intended impacts of a scientific breakthrough." — Jennifer Doudna
CRISPR Research at the IGI
At IGI, our goal is to use CRISPR and other advanced genomic technologies to tackle the world's greatest problems in health, the environment, and agriculture. We are especially interested in advancing research to meet the needs of underserved populations and ensuring equitable access to CRISPR-based advances.
You can learn more about our research programs on our program pages.
More Resources
Read the Book

A Crack in Creation takes the reader on a scientific journey, vividly exploring an unexpected discovery that has reshaped the future of humanity.
IGI Founder Jennifer Doudna wrote this popular science book about her personal and professional experiences in CRISPR research. Co-authored with her former graduate student, Sam Sternberg, A Crack in Creation offers a behind-the-scenes look at the development of CRISPR genome-editing technology, its applications, and ethical implications.
Watch the Documentary

The CRISPR story is filled with unexpected discoveries, eclectic characters, and provocative questions.
From executive producer Dan Rather and director Adam Bolt, Human Nature breaks down the science behind CRISPR, equipping and encouraging viewers to think about the potential implications of one of history's most transformative innovations. Watch on demand on Netflix or PBS. Visit the film's website for accompanying educational resources.