Genomics
Institute
Developing methods to precisely understand and control Cas9 and DNA repair outcomes
We are developing safer genome–editing tools that make only the exact DNA changes we need.
SHARE:
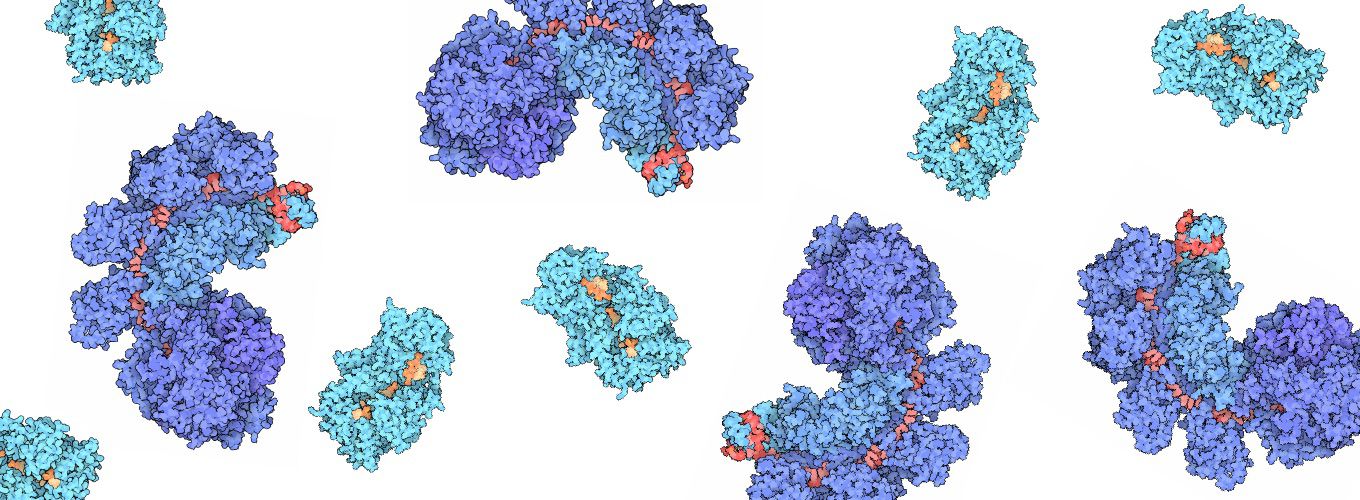
Our inability to precisely control either Cas9 or how cells repair Cas9-mediated breaks represents a huge roadblock that stands between us and the realization of CRISPR-based editing treatments for genetic disease. Cas9 can bind and cut at so-called "off-target" sites, potentially causing unintended genetic changes. Replacing a faulty gene with a healthy copy requires Cas9 to cut at its intended target and for the cell to repair the break by inserting exogenous DNA, but this outcome is very infrequent and experimenters can do little to affect the process. Developing methods to make Cas9 more accurate and to enhance the efficiency of gene insertion will be critical for eventually treating human genetic disease.
We study the steps that follow DNA cleavage by Cas9 to gain insight into how different repair pathways are initiated and how we might better control this process. Genome editing outcomes are currently unpredictable in part because we have fundamental gaps in our understanding of DNA repair. We are working to determine the molecular mechanisms by which cells process DNA damage, such as that which is incurred during genome editing, with the goal of furthering fundamental understanding of DNA repair and finding routes to high efficiency gene correction. Critically, technological advancements in gene replacement will not only benefit humans directly; they will also improve genome editing in plants, livestock, and microbes.
Share this project: