Advancing genome research for a better world
Advancing genome research for a better world
Founded by Nobel Laureate Jennifer Doudna, the Innovative Genomics Institute is using genome engineering to solve humanity’s greatest problems in health, climate, and sustainable agriculture.
Research Programs
 Human Health
Human Health
 Climate & Sustainable Agriculture
Climate & Sustainable Agriculture
 Advancing Genome Engineering
Advancing Genome Engineering
Resources
CRISPR Clinical Trials: A 2024 Update
Publications
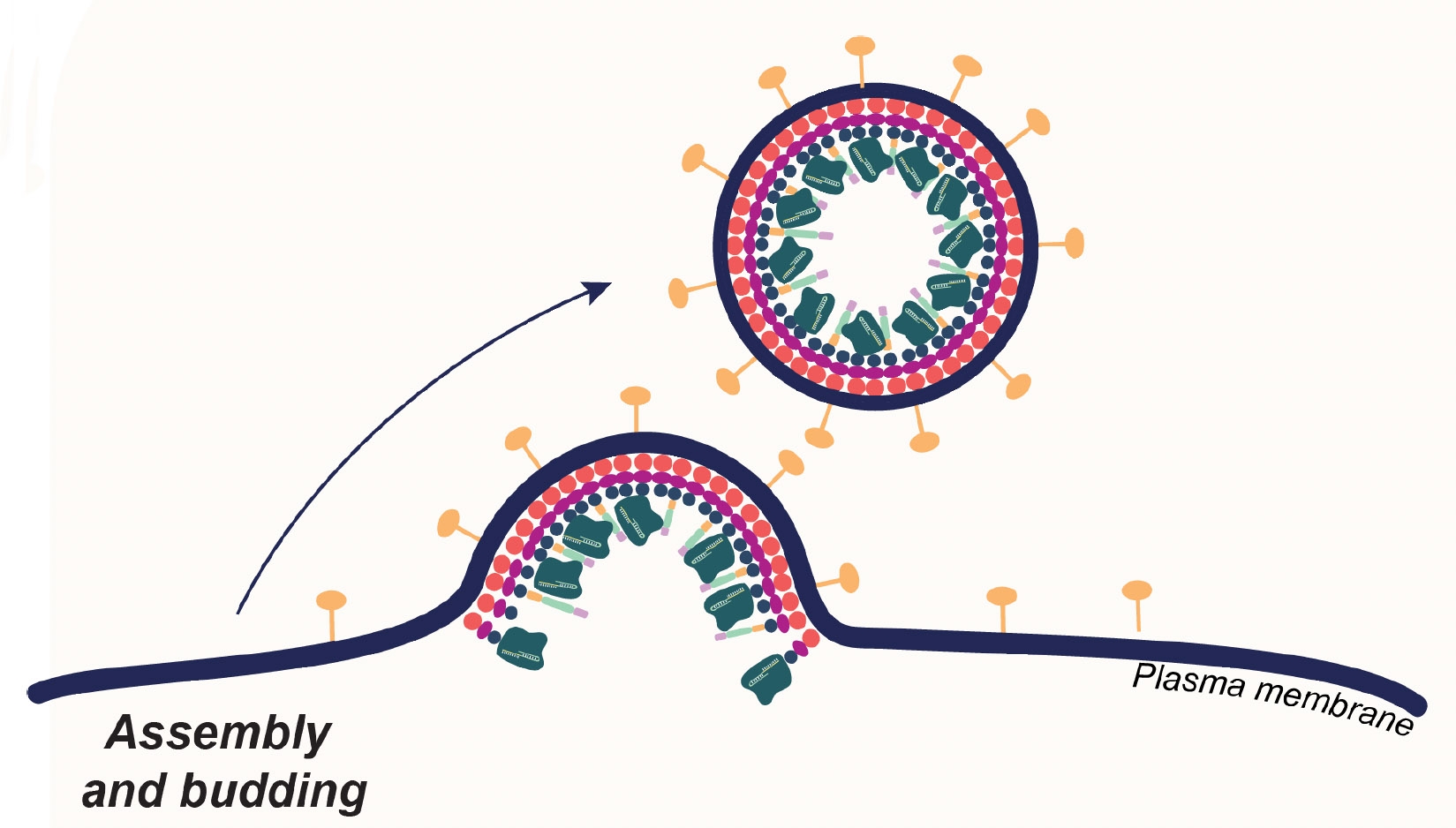
- Assembly of SARS-CoV-2 nucleocapsid protein with nucleic acid - Nucleic Acids Research
- Improving prime editing with an endogenous small RNA-binding protein - Nature
- A phage nucleus-associated RNA-binding protein is required for jumbo phage infection - Nucleic Acids Research
- CRISPR-based split luciferase as a biosensor for unique DNA sequences in situ - Methods in Molecular Biology
- Targeted nonviral delivery of genome editors in vivo - PNAS
- Engineering self-deliverable ribonucleoproteins for genome editing in the brain - Nature Communications
IGI in the News
- Could Lab-developed Tests Help Get CRISPR to Patients? - STAT News
- Gene-editing Pioneer Calls for Big Investment in CRISPR Technology - Financial Times
- Engineering the Microbiome: CRISPR Leads the Way - The Scientist
- Women in Enterprising Science Incubator is Giving Young Scientists Room to Grow - SF Business Times
- The FDA Approved The First CRISPR-Based Therapy. What’s Next? - NPR Science Friday
- Jennifer Doudna, Fyodor Urnov Want to Streamline Gene Editing Studies with Danaher's Help - Endpoints News